About these reports
Using data derived from Beacon Bispecific, The 2022 Bispecific Landscape Review series encompasses four reports that provide valuable insight to the 2022 bispecific landscape and beyond.
You will be given a detailed analysis of the clinical landscape (as of the 15th January 2023), along with a full list of notable 2022 regulatory announcements and deals, plus a look at what the future might hold for the bispecific space in 2023.
This four part series consists of:
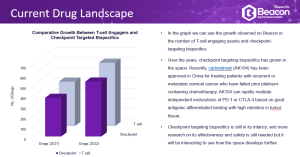
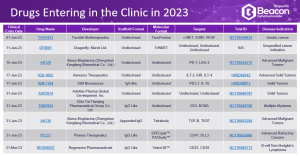
- The Drug Landscape – an extensive analysis of the drugs in the bispecific space
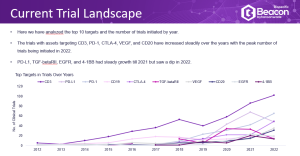
- The Trial Landscape – a deep dive into the 2022 trial landscape covering topics such as clinical trials initiated per year, phase distribution, recruitment status and disease indications
- The Future of Bispecific Therapies – take a sneak peek at the future of this space
- 2022 Regulatory Announcements – including key mergers and acquisitions, orphan drug designations and more
Find out more
Beacon is the essential decision-support tool for developers of complex therapeutics. Our market-defining, proprietary ontologies, combined with the most accurate and comprehensive life sciences data, provide our customers with unparalleled visibility of the drug and trial landscape.
With Beacon, you can make drug development decisions with confidence. This is why 23 of the world’s top 25 drug developers trust us.
Speak with our team
Our dedicated account and research teams can help you with questions by showing you how this insight and data is collated and leveraged on Beacon.


Beacon Bispecific
How the most complete bispecific database can help you
What we cover
This database solution is manually curated by sector-specific scientists and includes clinical trial and drug records for preclinical, active, approved, and discontinued antibody-based drugs that are designed to bind to two or more target antigens. We also cover:
- Whole Antibodies
- Antibody Fragments
- Augmented Antibodies
- Bispecific Fusion Proteins
How Beacon Bispecific works
Search the clinical trial and drug landscape by multiple targets, scaffold and over 80 different bispecific formats, instantaneously and/or extract the data points you need to conduct more complex analysis.