About these reports
Using data derived from Beacon ADC, The 2022 ADC Landscape Review series encompasses four reports that provide valuable insight to the 2022 ADC landscape and beyond.
You will be given a detailed analysis of the clinical landscape (as of the 15th January 2023), along with a full list of notable 2022 regulatory announcements and deals, plus a look at what the future might hold for the ADC space in 2023.
This four part series consists of:
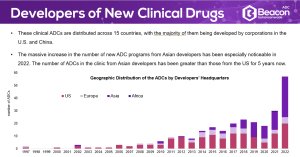
- The Clinical Landscape – an extensive analysis of the clinical ADC space.
- 2022 Regulatory Announcements – including approvals, fast track designations, breakthrough therapy designations, orphan drug designations, and more.
- ADC Deals in 2022 – a comprehensive list of all the ADC deals throughout 2022.
- What Does The Future Hold? – take a deep dive into late-stage ADCs in development and a look at new clinical ADCs in 2023.
Find out more
Beacon is the essential decision-support tool for developers of complex therapeutics. Our market-defining, proprietary ontologies, combined with the most accurate and comprehensive life sciences data, provide our customers with unparalleled visibility of the drug and trial landscape.
With Beacon, you can make drug development decisions with confidence. This is why 23 of the world’s top 25 drug developers trust us.
Speak with our team
Our dedicated account and research teams can help you with questions by showing you how this insight and data is collated on Beacon.



Beacon ADC
How the most complete ADC database can help you
What we cover
Beacon ADC is a manually curated database solution that includes clinical trial and drug records for preclinical, active, approved and discontinued:
- ADC (Antibody Drug Conjugates)
- Immunotoxins
- SMDCs (Small Molecule Drug Conjugates)
- Radioimmunoconjugates
- Ligand Conjugated Nanoparticles
- Liposomal Drug Conjugate and other comparable modalities
How Beacon ADC works
Search the trial and drug landscape by linker, payload and target alongside 20 other search criteria including disease, line of therapy, biomarker, sponsor etc. Instantly extract the data points you need to conduct more complex analysis.